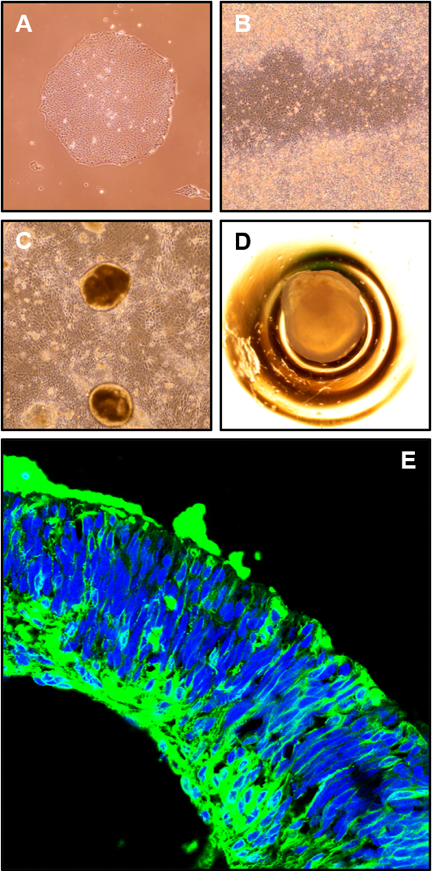
Stem Cell and Organoid Cultures
Our research focuses on disease modelling of Mendelian eye disorders by making use of induced pluripotent stem cells and genome editing. Diseases include monogenic retinal dystrophies (IRDs), which are generally progressive and may lead to blindness due to dysfunction and gradual loss of photoreceptors and/or supporting retinal pigment epithelial cells. This disease family is characterized by a significant genetic heterogeneity and clinical variability and involves more than 300 associated genes. The second group of diseases includes optic nerve hypoplasia (ONH), persistent hyperplastic primary vitreous (PHPV) and Norrie Disease (ND), the latter two of which are characterized by a maldevelopment of the neuroretina and retinal vasculature.
Disease-modelling is performed by using 3D retinal organoids, which are chemically differentiated from human induced pluripotent stem cells (iPSCs). In order to study the impact on vascular development, our retinal organoid model is complemented by endothelial cells differentiated from iPSC-derived mesodermal progenitors. Developmental and functional consequences of CRISPR/Cas9-mediated gene knockout or patient variant introduction in the organoid model is studied by morphologic and histologic techniques as well as gene expression approaches (mRNA sequencing (mRNA-seq) and single-cell RNA sequencing (scRNA-seq), where the latter technique provides the benefit of investigating individual subtypes of retinal cells. Other cell-based assays, including minigene assays for characterization of splicing and Luciferase reporter assays for detection of transcriptional activation, allow us to study novel genotype-phenotype associations and possibly lead to the identification of new therapeutic targets.
Diseases:
Norrie Disease
Optic Nerve Hypoplasia (ONH), atonal homolog 7 gene (ATOH7)
Methods:
CRISPR/Cas9 genome editing, next generation sequencing, iPSC-derived 3D retinal organoids, iPSC-derived mesodermal progenitors and endothelial cells, mRNA-seq, scRNA-seq, minigene assay, luciferase reporter assay
TEAM MEMBERS
Wolfgang Berger (PhD)
Silke Feil
David Grubich Atac (MD/PhD)
Samuel Koller (PhD)
Jordi Maggi (PhD)
Kevin Maggi
COLLABORATORS
Jane Sowden (PhD), Institute of Child Health, University College London, UK