Wagner Disease
Wagner disease (OMIM 143200) belongs to the group of vitreoretinopathy dieseases and is characterized by effects on multiple structures within the eye. The hallmark feature of this progressive disease is an apparently empty vitreous with fibrillary condensation, appearing as strands and veils. Patients can present cataract and mild to moderate myopia. The frequency of additional eye phenotypes increases with age. Among them are abnormal retinal vessel architecture, retinal thinning, chorioretinal atrophy and abnormal electroretinograms. No systemic abnormalities have been described. The disease was mapped to chromosome 15q13-14. Using the candidate gene sequencing approach we identified a novel splice site mutation in the VCAN gene (formerly CSPG2), which encodes an extra cellular matrix protein named versican. Depending on inclusion of exon 7 and 8 versican exists in 4 different isoforms, hence yielding varying numbers of attached glycosamino glycan (GAG) residues (three relevant variants are depicted below).
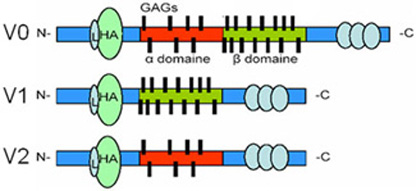
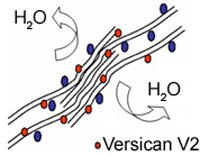
This splice site mutation causes drastic elevation of transcripts for the VCAN-V2 variant, where exon 8 has been spliced out. Interestingly, to date all reported mutations in VCAN leading to Wagner disease have been found in splice site donors and acceptors in introns 7 and 8, all leading to an over expression of variant 2.
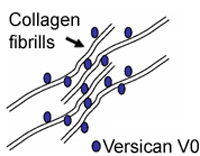
To explain the pathology of the splice mutation we are testing hypotheses of either haplo-insufficiency or disturbed isoform ratios by analyzing the expression of various VCAN transcript and protein isoforms. In the healthy vitreous, glycosaminoglycan-rich versican isoforms prevent clumping of collagen fibrills. Splicing defects in Wagner disease lead to drastic over expression of variant V2 carrying reduced attachment site for glycosamino glycans. This situation may lead to early liquefaction of the vitreous, as illustrated to the left.
We do offer genetic testing for Wagner disease by direct sequencing of the splice donor and acceptor sites of exons 7 and 8. Please refer to our genetic testing pages for more information on requirements for DNA diagnostics. Genetic Testing